Hence, the situation of losing 6 valance electrons doesn’t happen.Ĭonsider the element Chlorine (non-metal) This is because it is more energy efficient to gain 2 electrons than to lose 6 electrons. It will have the stable electron configuration of 2 as well. Note: You might wonder why doesn’t it lose 6 valence electrons.It will strive to gain 2 electrons to end up with an electron configuration of 2.8.It has the electron configuration of 2.6.It will strive to lose the 3 valence electrons to end up with an electron configuration of 2.8.It has the electron configuration of 2.8.3.It will strive to lose the solo valence electron to end up with an electron configuration of 2.It has the electron configuration of 2.1.Another way of interpreting this: Depending on the number of valence electrons, they will lose/gain different number of electrons. Hence, we say that a negative ion (anion) is formed.ĭepending on the position of the metal/non-metal on the periodic table, they will lose/gain different number of electrons. 
As they gain additional electrons, they will become negatively charged. Typically non-metal atoms gain valence electrons to achieve the noble gas configuration.Hence, we say that a positive ion (cation) is formed. As they lose the valence electrons, they will become positively charged. Typically, metal atoms lose their valence electrons to achieve the noble gas configuration.Hence, they will try to lose or gain electrons in order to fully fill their valence shell. Recall that atoms aim to achieve the stable noble gas configurations (having a fully filled valence shell).Ions are charged particles which are formed when an atom loses or gains electrons "As you go down the periodic table, the alkali metals become more inclined to lose their valence electron" and thus, "the amount of the element found in nature also decreases, later discovery dates.In ionic bonding, the participants are ions and they are kept together by the mutual force of attraction between oppositely charged ions. "All of these elements were first discovered in compounds some of the discoveries are hard to attribute due to the abundance and usage of the compounds," says Nataro. If an element is highly reactive, it's harder to find naturally. Since alkali metals are so reactive, they are usually found in conjunction with other metals in nature. So, all of the alkali metals like to make cations that have a charge of +1." When this happens, the atom is referred to as an ion and since it would have a positive charge, it is called a cation. "As electrons have a charge of -1, losing an electron causes the atom to have a charge of +1. Chip Nataro, chemistry professor at Lafayette College in Easton, Pennsylvania. All of the alkali metals like to give up their single valence electron," says Dr. In this process, the alkali metal is said to be oxidized, and whatever takes the electron from the alkali metal is reduced. "Since the alkali metals only have one valence electron, they typically achieve this state by giving up that electron. Noble gases (elements like neon and helium) are very unreactive because their outermost electron shells are full. In fact, reactivity in chemistry is defined by the number of electrons in the outermost shell. Having only one electron in the outermost shell makes it very easy for the atoms of alkali metals to reach points of stability – they just need to lose one electron! This willingness and ease of losing an electron to reach a state of equilibrium is known as high reactivity. This outermost shell is also called the valence shell, and the electrons that reside there are called valence electrons. However, elements in that first column of the periodic table all have one electron in their outermost shell. It's these shells of electrons and how alkali metals are structured that make them so reactive.Īll atoms naturally want to have a completely full outermost shell of electrons. The first shell can hold up to two electrons, the second up to eight, the third, 18 and the fourth, 32. These electrons exist in energy shells around the nucleus of the atoms, each of which can hold a varying number of electrons. Surrounding the nucleus of atoms are electrons, which are particles with a negative charge. 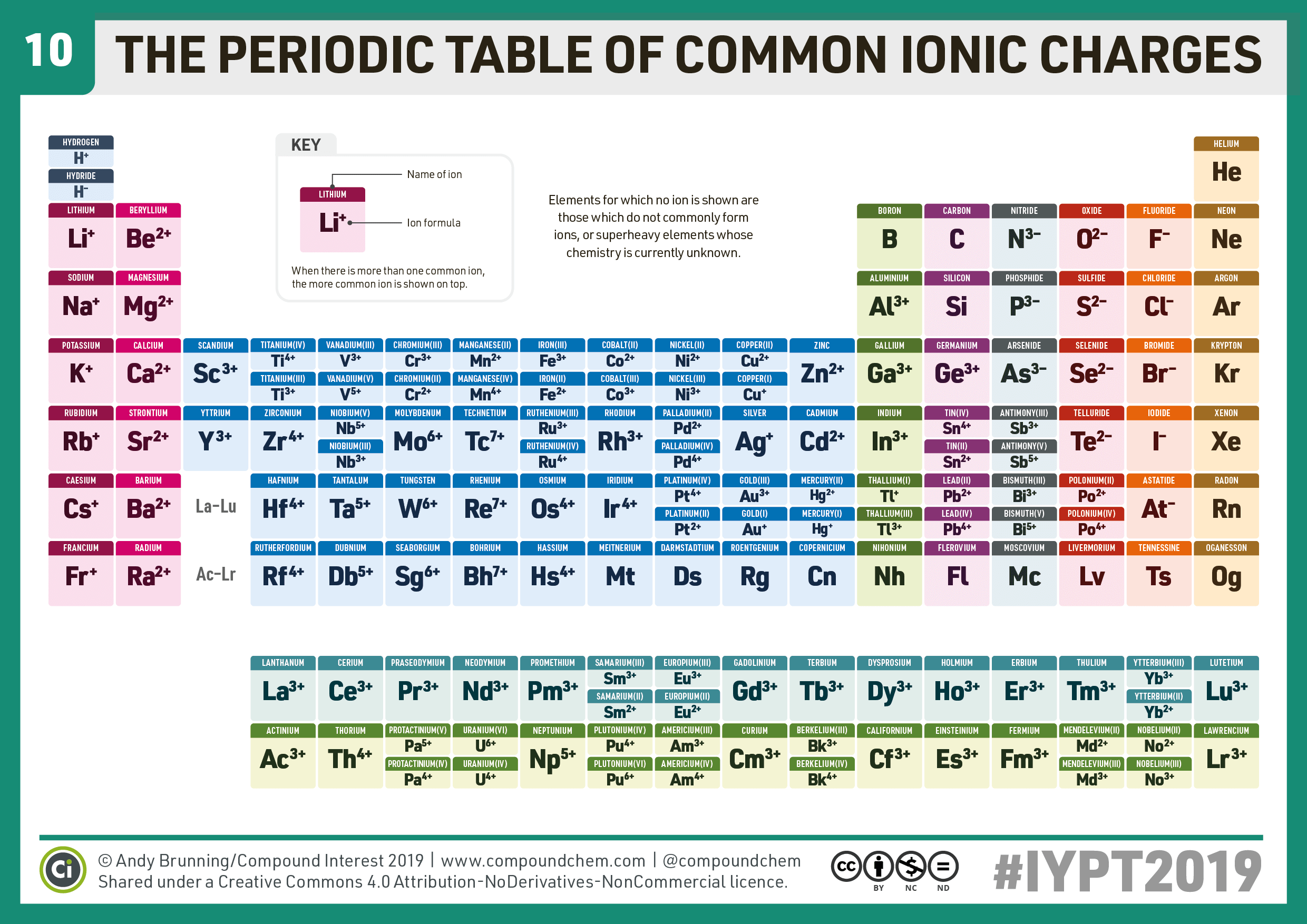
Substances that are highly alkaline can form strong bases able to neutralize acids and maintain a stable ph level.Įvery element has a nucleus, made up of protons and neutrons, and alkali metals are no different. Alkalinity refers to the pH of the substance, or the ability to neutralize acid. Alkali metals are so-called because when they react with water, they create highly alkaline substances. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
